Comprendre les microscopes et objectifs
Cette note correspond à la Section 9.1 du Guide de Ressources en Imagerie
Composants des microscopes | Concepts clés et spécifications | Exemples d'application de la microscopie optique

Un microscope est un dispositif optique utilisé pour reproduire un objet sur l'œil humain ou sur un appareil vidéo. Les premiers microscopes, composés de deux éléments, produisaient simplement une image plus grande de l'objet inspecté que ce que l'œil humain pouvait observer. La conception a évolué au cours de l'histoire du microscope pour intégrer aujourd'hui de multiples lentilles, filtres, polariseurs, séparateurs de faisceau, capteurs, sources d'éclairage et une multitude d'autres composants. Pour comprendre ces dispositifs optiques complexes, examinons les composants d'un microscope, les concepts et spécifications clés, ainsi que les applications.
Composants des microscopes
Un microscope composé est un microscope qui contient plusieurs éléments de lentille. Son fonctionnement est similaire à celui d'une simple loupe qui utilise une seule lentille pour grossir un petit objet afin que l'œil humain puisse en discerner les détails. Avec un grossisseur simple, l'objet est placé dans la distance focale de la lentille unique. Cela produit une image virtuelle agrandie. Avec un microscope, un système de lentilles relais remplace la lentille unique ; un objectif et un oculaire travaillent en tandem pour projeter l'image de l'objet sur l'œil, ou sur un capteur - selon l'application. Un microscope comporte deux parties qui augmentent le grossissement global du système : l'objectif et l'oculaire. L'objectif, situé le plus près de l'objet, transmet une image réelle de l'objet à l'oculaire. Cette partie du microscope est nécessaire pour produire le grossissement de base. L'oculaire, situé le plus près de l'œil ou du capteur, projette et agrandit cette image réelle et produit une image virtuelle de l'objet. Les oculaires produisent généralement un grossissement supplémentaire de 10X, mais cela peut varier de 1X à 30X. La Figure 1 illustre les composants d'un microscope composé. En outre, l'équation 1 montre comment calculer le grossissement global du système. Dans l'équation 1, m est le grossissement.

Figure 1 : Les composants d'un microscope composé.

Oculaires
Lorsque les microscopes ont été inventés, les oculaires ont joué un rôle majeur dans leur conception, car ils étaient le seul moyen de voir réellement l'objet inspecté. Aujourd'hui, des caméras analogiques ou numériques sont utilisées pour projeter une image de l'objet sur un moniteur ou un écran. Les oculaires de microscope se composent généralement d'une lentille de champ et d'une lentille d’œil, bien qu'il existe plusieurs conceptions qui créent chacune un champ de vision (FOV) plus large qu'une conception à élément unique. Pour un guide simple sur le choix du bon modèle, consultez la rubrique Choisir le bon oculaire.
Éclairage
L'éclairage d'un microscope est tout aussi important que le choix de l'oculaire ou de l' objectif approprié. Il est indispensable de choisir l'éclairage correct afin d'obtenir les résultats les plus concluants. Avant de décider du type d'éclairage à utiliser, il faut tenir compte de la configuration de l'application, de l'objet à inspecter et des résultats souhaités.
De nombreux microscopes utilisent un rétroéclairage plutôt qu'un éclairage traditionnel par lumière directe, car cette dernière sursature généralement l'objet à inspecter. Un type spécifique d'éclairage par rétroéclairage utilisé dans les applications de microscopie est l'éclairage de Köhler. Dans l'éclairage de Köhler, la lumière incidente d'une source d'éclairage, telle qu'une ampoule, inonde l'objet à inspecter d'une lumière provenant de l'arrière (Figure 2). Cet éclairage utilise deux lentilles convexes : la lentille collectrice et la lentille condenseur. Il est conçu pour fournir un éclairage brillant et uniforme sur le plan objet et sur le plan image où l'image produite par l'objectif est ensuite réimagée à travers l'oculaire. C'est important car cela permet de s'assurer que l'utilisateur n'imite pas le filament de l'ampoule. Comme le rétroéclairage inonde l'objet de lumière par l'arrière, il est également appelé éclairage à fond clair.
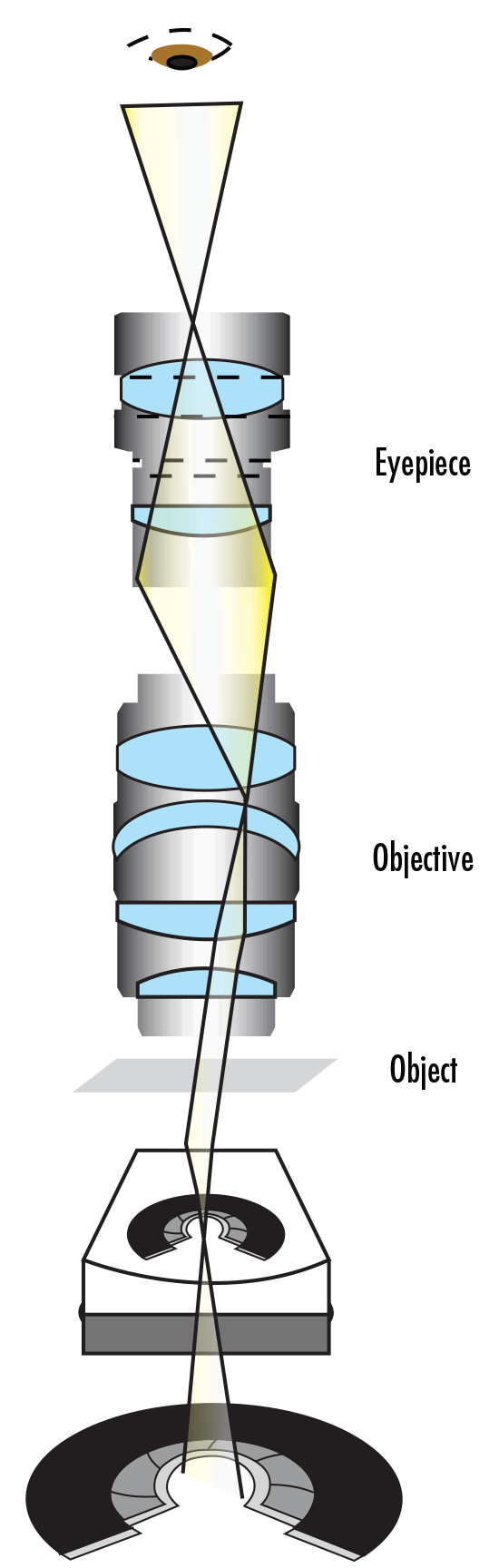
Figure 2 : Une configuration d'éclairage de Köhler.
L'éclairage à fond clair nécessite un changement d'opacité dans tout l'objet. Sans ce changement, l'éclairage crée un flou sombre autour de l'objet. Le résultat final est une image de contraste relatif entre les parties de l'objet et la source de lumière. Dans la plupart des cas, à moins que l'objet ne soit extrêmement transparent, l'image résultante permet à l'utilisateur de voir chaque partie de l'objet avec une certaine clarté ou résolution. Dans les cas où la transparence d'un objet rend difficile la distinction des caractéristiques en utilisant l'éclairage à fond clair, on peut utiliser l'éclairage à fond noir.
Avec l'éclairage à fond noir, les rayons lumineux directs ne sont pas envoyés dans l'objectif mais frappent l'objet à un angle oblique. Il est important de garder à l'esprit que ces rayons éclairent toujours l'objet dans le plan de l'objet. L'image qui résulte de l'éclairage à fond noir produit un contraste élevé entre l'objet transparent et la source lumineuse. Lorsqu'il est utilisé dans une installation de microscopie, l'éclairage à fond noir produit une source lumineuse qui forme un cône de lumière inversé bloquant les rayons centraux de la lumière mais permettant aux rayons obliques d'éclairer l'objet. La Figure 3 illustre un exemple de configuration d'éclairage à fond noir où le cône creux de lumière est l'ouverture numérique de l'objectif. En comparaison, aucun rayon n'est bloqué dans une configuration d'éclairage à fond clair. La conception de l'éclairage à fond noir oblige la lumière à éclairer l'objet inspecté, mais pas à pénétrer dans le système optique, ce qui est préférable pour un objet transparent.

Figure 3 : Configuration de l’éclairage à fond noir.
Un troisième type d'éclairage utilisé en microscopie est l'épi-éclairage. L'épi-éclairage produit de la lumière au-dessus de l'objectif. Par conséquent, l'objectif et la source d'épi-éclairage se substituent à une configuration d'éclairage de Köhler. L'utilisation de l'objectif pour une grande partie de l'éclairage rend l'épi-éclairage très compacte – un avantage majeur de cette conception. La Figure 4 illustre une configuration d'épi-éclairage fréquemment utilisée dans les applications de fluorescence. Pour plus d'informations sur la microscopie de fluorescence, Voir « Fluorophores et filtres pour la microscopie à fluorescence ».

Figure 4 : Une installation d'épi-éclairage

Objectifs :
Les objectifs permettent aux microscopes de fournir des images agrandies et réelles et sont, peut-être, le composant le plus complexe d'un système de microscope en raison de leur conception multi-éléments. Les objectifs sont disponibles avec des grossissements allant de 2X à 200X. Ils sont classés en deux grandes catégories : le type réfractif traditionnel et le type réfléchissant. Chaque catégorie est encore divisée en types : conjugué fini et conjugué à l’infini (corrigé à l'infini). Afin de choisir le bon objectif, il est important de connaître les avantages d'une catégorie et d'un type par rapport à un autre.
Objectifs : Réfractif
La catégorie d'objectifs la plus couramment utilisée est la catégorie réfractive. Dans une conception réfractive, la lumière qui traverse le système est réfractée, ou pliée, par les éléments optiques. Chaque élément optique est généralement doté d’un traitement antireflets afin de réduire les réflexions arrière et d'améliorer le débit lumineux global. Les objectifs réfractifs sont souvent utilisés dans les applications de vision industrielle qui nécessitent la résolution de détails extrêmement fins. Il existe de nombreux modèles d'objectifs réfractifs, chacun utilisant des configurations optiques différentes. Les conceptions peuvent aller de deux éléments dans les objectifs achromatiques de base (une lentille achromatique et une lentille ménisque) à quinze éléments dans les objectifs plan-apochromatiques (Figure 5). Les objectifs plan-apochromatiques sont les objectifs haut de gamme les plus complexes, avec une correction chromatique et de champ plat effectuée dans l'objectif lui-même.
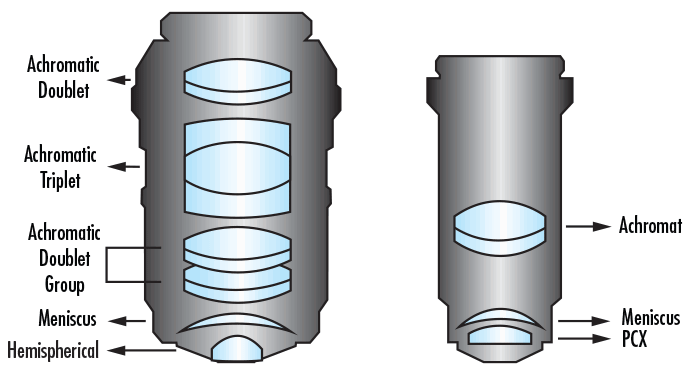
Figure 5 : Modèle d’objectif apochromatique (à gauche) contre modèle d’objectif achromatique (à droite).
Objectifs : Réfléchissant
Les objectifs réfléchissants utilisent un design réfléchissant ou basé sur un miroir. Ils sont souvent méconnus par rapport à leurs homologues réfractifs, bien qu'ils puissent corriger de nombreux problèmes présents dans ces derniers. Les objectifs réfléchissants sont constitués d'un système de miroirs primaire et secondaire (Figure 6) permettant de grossir et de relayer l'image de l'objet inspecté. Edmund Optics® utilise la conception populaire de Schwarzschild, bien que d'autres conceptions soient disponibles. Étant donné que la lumière est réfléchie par les surfaces métalliques et non réfractée par les surfaces en verre, les objectifs réfléchissants ne souffrent pas des mêmes aberrations que les objectifs réfractifs et, par conséquent, ne nécessitent pas de conceptions supplémentaires pour compenser ces aberrations. Les objectifs réfléchissants peuvent produire une meilleure efficacité lumineuse ainsi qu'un meilleur pouvoir de résolution pour l'imagerie de détails fins, car le système dépend principalement du traitement du miroir plutôt que du substrat en verre utilisé. Un autre avantage des objectifs réfléchissants est la possibilité de travailler plus profondément dans les régions spectrales de l'ultraviolet (UV) ou de l'infrarouge (IR) grâce à l'utilisation de miroirs par rapport aux composants optiques réfractifs conventionnels.

Figure 6 : L'anatomie d'un objectif réfléchissant.
Concepts clés et spécifications
La plupart des spécifications des objectifs de microscope sont indiquées sur le corps de l'objectif lui-même : conception/norme de l'objectif, grossissement, ouverture numérique, distance de travail, distance entre l'objectif et l'image et correction de l'épaisseur de la lamelle. La Figure 7 montre comment lire les spécifications d'un objectif de microscope. Comme les spécifications sont situées directement sur le corps de l'objectif, il est facile de savoir exactement ce que l'on a, un fait très important lorsqu'on incorpore plusieurs objectifs dans une application. Toutes les autres spécifications, telles que la distance focale, le champ de vision et la longueur d'onde de conception, peuvent être facilement calculées ou trouvées dans les spécifications fournies par le vendeur ou le fabricant.

Figure 7 : Un objectif de microscope typique à transmission.
La norme des objectifs
Si l'objectif suit une norme de microscope simple (telle que DIN ou JIS), elle est indiquée sur le corps pour montrer quelles spécifications requises doivent être présentes dans le système. La plupart des microscopes composés utilisent la norme Deutsche Industrie Norm, ou DIN. La norme DIN prévoit une distance de 160 mm entre la bride de l'objectif et la bride de l'oculaire (Figure 8). L'autre norme disponible est la norme industrielle japonaise, ou JIS. La norme JIS prévoit une distance de 170 mm entre la bride de l'objectif et la bride de l'oculaire (Figure 9). Il est nécessaire de prêter attention à ces deux distances lors du choix de l'objectif et de l'oculaire appropriés, afin de s'assurer que l'image projetée par le premier est correctement reproduite par le second. Bien que les distances d'image soient différentes pour les normes DIN et JIS, il n'y a pas de différence dans les performances optiques ; elles sont de qualité égale. De même, chaque norme utilise le même filetage de montage RMS de 0,7965" x 36 TPI.
Les normes DIN et JIS ont toujours été utilisées pour les microscopes composés classiques. Certains fabricants de microscopes préfèrent répertorier la longueur de la lentille tube en fonction des propriétés optiques plutôt que des propriétés mécaniques. Pour un objectif standard DIN, la longueur de la lentille tube passe à 150 mm car l'oculaire représente le plan d'image intermédiaire (Figure 8). Enfin, il existe une dimension généralement indiquée pour les objectifs afin de permettre à l'utilisateur de savoir systématiquement de quelle longueur il s'agit : la distance parfocale (DP). La distance parfocale est la distance entre la bride de l'objectif et l'objet à inspecter. Cette distance est de 45 mm pour les objectifs DIN et de 36 mm pour les objectifs JIS (Figures 8 et 9).
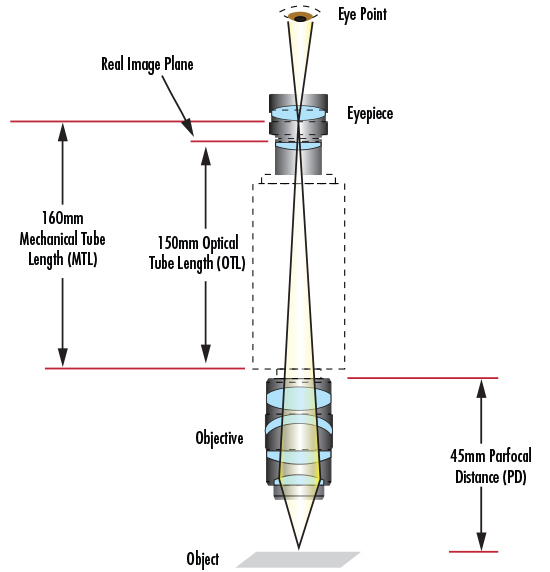
Figure 8 : La norme DIN.
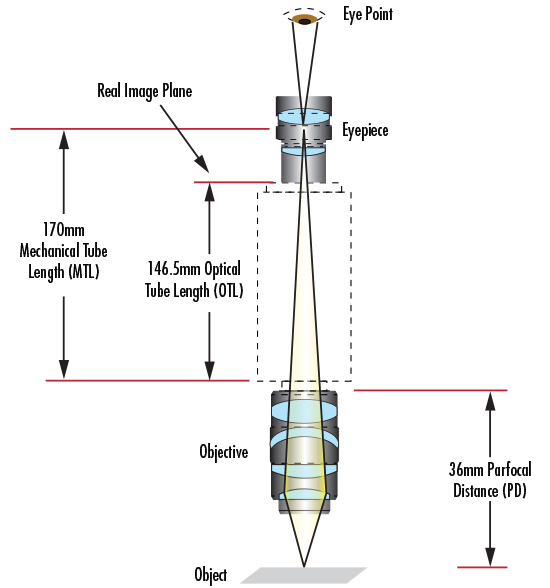
Figure 9 : La norme JIS.
Grossissement
Les oculaires et les objectifs ont tous deux un grossissement qui contribue au grossissement global du système. Le grossissement est généralement indiqué par un X à côté d'une valeur numérique. La plupart des objectifs comportent une bande colorée sur toute la circonférence du corps qui indique leur grossissement (Figure 7). Par exemple, une bande jaune indique un grossissement de 10X.
Ouverture numérique
L'ouverture numérique (NA) d'un objectif est fonction de la distance focale et du diamètre de la pupille d'entrée. Les objectifs à grande NA nécessitent parfois l'utilisation d'huiles d'immersion entre l'objet inspecté et l'avant de l'objectif. En effet, la NA la plus élevée pouvant être atteinte dans l'air est un NA de 1 (correspondant à un angle de lumière de 90°). Pour obtenir un angle plus grand et augmenter la quantité de lumière entrant dans l'objectif (équation 2), il est nécessaire d'utiliser de l'huile d'immersion (indice de réfraction généralement = 1,5) pour modifier l'indice de réfraction entre l'objet et l'objectif. Les objectifs à haute NA associés à l'huile d'immersion constituent une alternative simple au changement d'objectif, qui peut s'avérer coûteux.
Champ de vision
Le champ de vision ou FOV est la zone de l'objet qui est imagée par un système de microscope. La taille du champ de vision est déterminée par le grossissement de l'objectif. Lorsqu'on utilise un système oculaire-objectif, le champ de vision de l'objectif est agrandi par l'oculaire pour l'observation. Dans un système caméra-objectif, ce champ de vision est relayé sur un capteur de caméra. Le capteur d'une caméra est rectangulaire et ne peut donc prendre des images que sur une partie du champ de vision circulaire de l'objectif. En revanche, la rétine de votre œil peut prendre des images d'une zone circulaire et capture la totalité du champ de vision. C'est pourquoi le champ de vision produit par un système caméra-microscope est généralement légèrement plus petit que celui d'un système oculaire-microscope. Les équations 3 et 4 peuvent être utilisées pour calculer le champ de vision dans les systèmes susmentionnés. Dans les équations 3 et 4, $ \small{H_{\small{\text{Capteur caméra}}}} $ est la taille du capteur de la caméra et $ \small{H_{\small{\text{Diaphragme champ oculaire}}}} $ est le diaphragme de l'oculaire.
Épaisseur de la lamelle
Lors de l'observation de matériaux fluides tels que des bactéries, des cultures cellulaires, du sang, etc., il est nécessaire d'utiliser une lamelle couvre-objet afin de protéger l'objet inspecté et les composants du microscope de toute contamination. Une lamelle couvre-objet, ou lame de verre pour microscope, modifie la façon dont la lumière se réfracte de l'objet vers l'objectif. Par conséquent, l'objectif doit effectuer des corrections optiques appropriées pour produire une image de la meilleure qualité. C'est pourquoi les objectifs indiquent une gamme d'épaisseurs de lamelles pour lesquelles ils sont optimisés. Généralement, cette valeur est indiquée après le symbole de l'infini (qui indique qu'un objectif est un conjugué à l’infini ou un modèle corrigé à l'infini) et va de zéro (pas de correction de la lamelle couvre-objet) à 0,17 mm.
Correction de la qualité
La qualité d'un objectif et d'un oculaire détermine la performance du système. Outre le choix du grossissement et de la complexité du dessin, la compréhension de la correction de la qualité est extrêmement importante pour décider du type d'objectif à utiliser. La correction de la qualité (achromatique, apochromatique, plane, semi-plane) est indiquée sur l'objectif lui-même pour permettre à l'utilisateur de voir facilement la conception de l'objectif en question. Il existe généralement deux niveaux de correction d'aberrations chromatiques : achromatique et apochromatique. Les objectifs achromatiques font partie des objectifs les plus simples et les moins chers. Ils sont conçus pour corriger l'aberration chromatique dans les longueurs d'onde rouge et bleue, en plus d'être corrigés pour l'aberration sphérique dans une longueur d'onde verte. La correction limitée de l'aberration chromatique et l'absence d'un champ de vision plat réduisent les performances de l'objectif. Les objectifs apochromatiques, en revanche, offrent une plus grande précision et sont corrigés chromatiquement pour le rouge, le bleu et le jaune. Ils offrent également une correction de l'aberration sphérique pour une large gamme spectrale et ont généralement une longue distance de travail étant donné les ouvertures numériques (NA) extrêmement élevées que cette conception optique offre. Les objectifs apochromatiques sont idéaux pour les applications en lumière blanche, tandis que les objectifs achromatiques conviennent mieux aux applications monochromatiques. Cependant, les deux modèles d'objectifs souffrent considérablement de la distorsion et de la courbure de champ, qui s'aggravent à mesure que le grossissement de l'objectif augmente. Par conséquent, il est toujours important de se concentrer sur les performances complètes du système, plutôt que sur les seules performances de l’objectif.
Les objectifs plans, également appelés semi-plans ou microplans, corrigent la courbure de champ. La courbure de champ est un type d'aberration présent lorsque l'image hors axe ne peut être mise au point dans un plan d'image plat, ce qui donne une image floue lorsqu'elle s'écarte de l'axe optique. La Figure 10 illustre la planéité du champ mesurée radialement à partir du centre pour des objectifs achromatiques, semi-plans et plans. Les objectifs achromatiques ont un champ plat dans les 65% centraux de l'image. Les objectifs plans ont la meilleure correction globale et affichent mieux que 90% du champ plat et focalisé. Les objectifs semi-plans sont intermédiaires par rapport aux deux autres types, 80% du champ apparaissant plat.

Figure 10 : Correction du champ plat : achromatique 65% (gauche) vs. semi-plan 80% (centre) vs. plan 90% (droite).
Les objectifs en fluorite corrigent davantage les aberrations en utilisant des types de verre avancés contenant du spath fluor ou d'autres substituts synthétiques. Tout comme les objectifs achromatiques, les objectifs en fluorite sont conçus pour corriger les aberrations chromatiques pour les longueurs d'onde rouges et bleues. Cependant, les objectifs en fluorite sont conçus pour corriger l'aberration sphérique à deux ou trois longueurs d'onde au lieu de la seule longueur d'onde verte. Ils ont généralement une NA plus élevée, et présentent un meilleur pouvoir de résolution et un degré de contraste plus élevé.
Conjugué Fini
Dans une conception optique de conjugaison finie, la lumière d'une source (non à l'infini) est focalisée en un point (Figure 11). Dans le cas d’un microscope, l'image de l'objet inspecté est agrandie et projetée sur l'oculaire, ou le capteur si l'on utilise une caméra. La distance particulière à travers le système est caractérisée par la norme DIN ou JIS ; tous les microscopes à conjugaison finie suivent l'une de ces deux normes. Ces types d'objectifs représentent la majorité des microscopes de base. Les modèles conjugués finis sont utilisés dans les applications où le coût et la facilité de conception sont des préoccupations majeures.

Figure 11 : Une conception simplifiée du microscope à conjugaison finie.
Conjugué à l’infini (corrigé à l’infini)
Dans une conception optique de conjugaison finie, la lumière d'une source placée à l'infini est focalisée en un petit point. Dans un objectif, le point est l'objet à inspecter et l'infini pointe vers l'oculaire, ou vers le capteur si l'on utilise une caméra (Figure 12). Ce type de conception moderne utilise une lentille tube supplémentaire entre l'objet et l'oculaire afin de produire une image. Bien que cette conception soit beaucoup plus compliquée que sa contrepartie conjuguée finie, elle permet d’introduire des composants optiques tels que des filtres, des polariseurs et des séparateurs de faisceau dans le chemin optique. Il est ainsi possible d'effectuer des analyses d'images et des extrapolations supplémentaires dans des systèmes complexes. Par exemple, l'ajout d'un filtre entre l'objectif et la lentille tube permet d'observer des longueurs d'onde spécifiques de la lumière ou de bloquer les longueurs d'onde indésirables qui pourraient autrement interférer avec la configuration. Les applications de microscopie à fluorescence utilisent ce type de conception. Un autre avantage de l'utilisation d'une conception conjuguée infinie est la possibilité de varier le grossissement en fonction des besoins spécifiques de l'application. Le grossissement de l'objectif étant le rapport entre la distance focale de la lentille tube $ ( \small{f_{\small{\text{Lentille tube}}}} ) $ et la distance focale de l'objectif $ (\small{f_{\small{\text{Objectif}}}} ) $(équation 5), une augmentation ou diminution de la distance focale de la lentille tube modifie le grossissement de l'objectif. En général, la lentille tube est une lentille achromatique avec une distance focale de 200 mm, mais d'autres distances focales peuvent également être substituées, ce qui permet de personnaliser le grossissement total d'un système de microscope. Si un objectif est conjugué à l'infini, il y aura un symbole d'infini situé sur le corps de l'objectif.

Figure 12 : Conception simplifiée d’un microscope à conjugaison infinie (corrigé à l’infini).
Examples d’application de microscopie optique
Afin de comprendre comment les composants d'un microscope peuvent être combinés à divers produits optiques, d'imagerie et photoniques, considérons les applications de microscopie optique suivantes : la microscopie à fluorescence et l'ablation laser. Chacun d'entre eux utilise sa propre configuration afin de travailler avec les composants d'un microscope.
Microscopie de fluorescence
Un fluorophore (ou colorant fluorescent) est utilisé pour marquer les protéines, les tissus et les cellules à des fins d'examen ou d'étude. Les fluorophores peuvent absorber la lumière d'une longueur d'onde et émettre (fluorescer) une lumière d'une autre longueur d'onde. Dans une configuration typique de microscopie à fluorescence, trois filtres sont utilisés : un filtre d'excitation, un filtre d'émission et un filtre dichroïque. Chaque fluorophore a une bande de longueur d'onde d'absorption ou d'excitation spécifique, le filtre d'excitation ne transmettra que cette gamme spécifique de longueurs d'onde. Le fluorophore, une fois excité, émettra une gamme différente de longueurs d'onde. Le filtre d'émission ne transmet que les longueurs d'onde d'émission. Un filtre dichroïque spécialement conçu pour réfléchir les longueurs d'onde d'émission et transmettre les longueurs d'onde d'excitation est utilisé pour séparer les canaux d'excitation et d'émission. La Figure 13 illustre une configuration typique d'imagerie par fluorescence. Pour plus d'informations sur la microscopie à fluorescence, consultez la rubrique Fluorophores et filtres optiques pour la microscopie à fluorescence.

Figure 13 : Une configuration typique de microscope à fluorescence.
Ablation au laser
Deux utilisations courantes de lasers sont (1) le chauffage d'un matériau sur une base ou (2) l'ablation d'un matériau d'une base. Les systèmes d'ablation par laser nécessitent des composants de microscope en raison de la précision de la manipulation du faisceau (c'est-à-dire la focalisation, la courbure, la réduction de la dispersion, etc. Une installation d'ablation au laser contient généralement des optiques personnalisées plutôt que des composants standard, le laser étant précisément conçu dans le système (Figure 14). Le laser est orienté en épi-éclairage afin d'utiliser la capacité de l'objectif du microscope à focaliser la lumière sur le plan de l'objet, et de produire des spots de taille extrêmement réduite avec des aberrations minimales. De plus, un oculaire permet à l'utilisateur de voir où se trouve le laser et de s'assurer que tout fonctionne correctement. Des filtres sont nécessaires pour empêcher le laser de causer des dommages à l'œil de l'utilisateur. Les installations d'ablation laser sont utilisées dans les applications médicales et biologiques car elles offrent une plus grande précision que les méthodes chirurgicales conventionnelles.
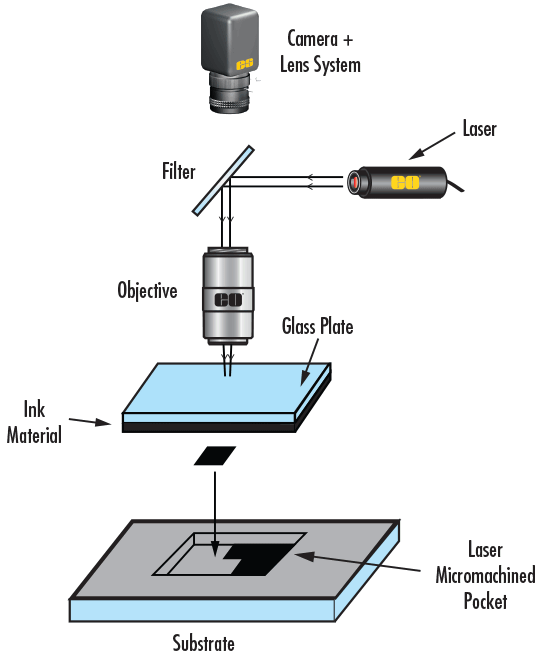
Figure 14 : Une installation typique d'ablation laser.
Le microscope et les objectifs sont des systèmes optiques complexes aux multiples usages. Ils ne sont plus uniquement utilisés pour des applications biologiques (par exemple, pour examiner des cellules de joue dans un cours d'introduction à la biologie), mais plutôt pour étudier la longueur d'onde d'émission d'un flourophore, pour analyser un défaut de 5 μm sur une pièce usinée, pour superviser l'ablation de matériau sur une base et dans le cadre d'une multitude d'autres applications dans les secteurs de l'optique, de l'imagerie et de la photonique. Comprendre l'importance de chaque élément constitutif d'un microscope et leurs spécifications permet à tout utilisateur de choisir le meilleur système et d'obtenir les meilleurs résultats.
sont généralement de l'ordre d'un pixel ou moins.












 Section précédente
Section précédente















ou consulter les numéros d’autres pays
facile à utiliser
entrer les numéros de stock pour commencer
Copyright 2023 | Edmund Optics, Ltd Unit 1, Opus Avenue, Nether Poppleton, York, YO26 6BL, UK
POLITIQUE DE CONFIDENTIALITÉ | POLITIQUE DE COOKIES | CONDITIONS GÉNÈRALES | CONDITIONS GÉNÈRALES B2C | MENTIONS LÉGALES | ACCESSIBILITÉ
L'entreprise Edmund Optics GmbH en Allemagne agit comme un mandataire d'Edmund Optics BV aux Pays-Bas.
Le titulaire du contrat est Edmund Optics BV aux Pays-Bas.
Ce contenu peut comporter des éléments générés ou modifiés à l'aide de l'intelligence artificielle (IA).
The FUTURE Depends On Optics®